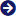Quinine Sulphate
5800 INR/Kilograms
Product Details:
- Structural Formula C20H24N2O2S
- Boiling point Not applicable (decomposes at high temperature)
- EINECS No 205-735-7
- Shelf Life 3 years
- Melting Point 250C (decomposes)
- Storage Store below 30C in a dry, well-ventilated area, away from light
- Heavy Metal (%) Max 0.001%
- Click to view more
X
Quinine Sulphate Price And Quantity
- 25 Kilograms
- 5800 INR/Kilograms
- +255° to +265° (C=1, H2SO4)
- Complies with IP/BP/USP
- Approx. 0.4 g/cm³
- Handle with suitable protective equipment
- Non-hazardous as per IATA/DGR
- HDPE drums, 25kg
- Available on request
- Pharmaceuticals, research and laboratory testing
- Complies with BP/USP Specifications
- Not less than 98% and not more than 102%
Quinine Sulphate Product Specifications
- Soluble in water, sparingly soluble in ethanol
- API/Pharmaceutical Raw Material
- Neutral
- 398.49 g/mol
- Very bitter
- White crystalline powder
- Micronized
- 205-735-7
- C20H24N2O2S
- Not applicable (decomposes at high temperature)
- 99% min
- 6119-70-6
- Max 0.001%
- Store below 30C in a dry, well-ventilated area, away from light
- 3 years
- 250C (decomposes)
- Yes, toxic in large doses
- Odorless
- Solid
- 29392090
- Quinine Sulphate
- Pharma Grade
- C20H24N2O2S
- Used in the treatment of malaria and nocturnal leg cramps
- Not more than 0.5%
- White
- Quinine Sulphate
- +255° to +265° (C=1, H2SO4)
- Complies with IP/BP/USP
- Approx. 0.4 g/cm³
- Handle with suitable protective equipment
- Non-hazardous as per IATA/DGR
- HDPE drums, 25kg
- Available on request
- Pharmaceuticals, research and laboratory testing
- Complies with BP/USP Specifications
- Not less than 98% and not more than 102%
Product Description
Established in the year 2000, we are emerging as one of the trusted companies engaged in formulating and supplying Quinine Sulphate. It is most commonly prescribed for malaria due to its ability to kill the malaria parasite residing inside red blood cells. This medicinal compound also finds its use for the treatment and prevention of nocturnal leg cramps. Quinine Sulphate is known to have molecular formula C40H50N4O8S with a molar mass of 746.92 g/mol.
Features:
- It is an alkaloid
Derived from the bark of the cinchona tree
- Have mild antipyretic & analgesic properties
Product Details:
- Product: Quinine Sulphate
- CAS number: 6119-70-6
- Grade: IP/BP/EP
- Description: White Dry Powder.
- Empirical formula: (C20H24N2O2)2.H2SO4
- Mol. wt.: 782.95 gms/mole
- Assay: NLT 99% & NMT 101% on /kaival-impex/dry basis.
- LOD: 5%(NMT)
- Solubility: Slightly soluble in water, Springily soluble in boiling water and alcohol.
- pH: Between 5.7 to 6.6
- Specific optical-Rotation: Between -2370 to -2450
- Sulphated ash: 0.1%(NMT)
Premium Pharma Grade Standards
Our Quinine Sulphate meets stringent BP/USP/IP specifications, ensuring high quality and pharmaceutical compliance. With an assay range of not less than 98% and not more than 102%, it guarantees reliable performance in medical and laboratory applications. Detailed certificates of analysis are available upon request for complete traceability and peace of mind.
Versatile Applications and Benefits
Quinine Sulphates primary applications include the treatment of malaria and management of nocturnal leg cramps. Its consistent micronized particle size offers optimal solubility and effectiveness in pharmaceutical formulations. It is also widely used in research and laboratory testing, providing valuable support in scientific studies and drug development.
Safe Handling and Reliable Packaging
This product is packaged in robust 25kg HDPE drums to ensure safe transit and storage. It is classified as non-hazardous for air and land transport as per IATA/DGR guidelines. For best results and longevity, store below 30C in a dry, well-ventilated space, away from direct light, and always use suitable protective equipment during handling.
FAQs of Quinine Sulphate:
Q: How should Quinine Sulphate be stored to maintain its efficacy?
A: Store Quinine Sulphate in its original HDPE drum, below 30C in a dry and well-ventilated area, away from direct light to preserve its stability and effectiveness.Q: What benefits does Quinine Sulphate offer in pharmaceutical applications?
A: Quinine Sulphate is highly valued for its efficacy in treating malaria and nocturnal leg cramps, supported by its high purity (>99%) and compliance with international pharmacopeial standards, ensuring safety and consistency in medicinal use.Q: When should a Certificate of Analysis for Quinine Sulphate be requested?
A: A Certificate of Analysis is available upon request and is typically required before bulk purchase, quality validation, or regulatory submissions to confirm the products compliance and specifications.Q: Where is Quinine Sulphate commonly used aside from pharmaceutical manufacturing?
A: Besides its use in medicines, Quinine Sulphate is also widely utilized in research and laboratory testing for analytical and scientific studies.Q: What safety measures are recommended when handling Quinine Sulphate?
A: Always handle Quinine Sulphate using suitable protective equipment such as gloves and goggles, as it is toxic in large doses and can be hazardous upon direct contact or inhalation.Q: How is the quality of Quinine Sulphate ensured during transportation?
A: Quinine Sulphate is packaged in secure 25kg HDPE drums, classified as non-hazardous for air transport (IATA/DGR), ensuring safe delivery and compliance with shipping regulations.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email